Marine chemistry, also known as ocean chemistry or chemical oceanography, is influenced by plate tectonics and seafloor spreading, turbidity currents, sediments, pH levels, atmospheric constituents, metamorphic activity, and ecology. The field of chemical oceanography studies the chemistry of marine environments including the influences of different variables. Marine life has adapted to the chemistries unique to earth's oceans, and marine ecosystems are sensitive to changes in ocean chemistry.
The impact of human activity on the chemistry of the earth's oceans has increased over time, with pollution from industry and various land-use practices significantly affecting the oceans. Moreover, increasing levels of carbon dioxide in the earth's atmosphere have led to ocean acidification, which has negative effects on marine ecosystems. The international community has agreed that restoring the chemistry of the oceans is a priority, and efforts toward this goal are tracked as part of Sustainable Development Goal 14.
Chemical oceanography is the study of the chemistry of Earth's oceans. An interdisciplinary field, chemical oceanographers study the distributions and reactions of both naturally occurring and anthropogenic chemicals from molecular to global scales.[2]
Due to the interrelatedness of the ocean, chemical oceanographers frequently work on problems relevant to physical oceanography, geology and geochemistry, biology and biochemistry, and atmospheric science. Many chemical oceanographers investigate biogeochemical cycles, and the marine carbon cycle in particular attracts significant interest due to its role in carbon sequestration and ocean acidification.[3] Other major topics of interest include analytical chemistry of the oceans, marine pollution, and anthropogenic climate change.
Organic compounds in the oceans
Colored dissolved organic matter (CDOM) is estimated to range 20-70% of carbon content of the oceans, being higher near river outlets and lower in the open ocean.[4]
Marine life is largely similar in biochemistry to terrestrial organisms, except that they inhabit a saline environment. One consequence of their adaptation is that marine organisms are the most prolific source of halogenated organic compounds.[5]
Chemical ecology of extremophiles
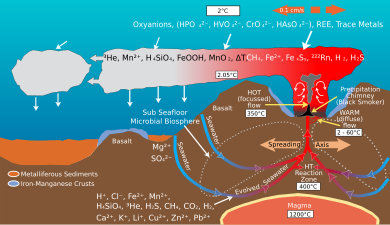
The ocean provides special marine environments inhabited by extremophiles that thrive under unusual conditions of temperature, pressure, and darkness. Such environments include hydrothermal vents and black smokers and cold seeps on the ocean floor, with entire ecosystems of organisms that have a symbiotic relationship with compounds that provide energy through a process called chemosynthesis.
Plate tectonics

Seafloor spreading on mid-ocean ridges is a global scale ion-exchange system.[6] Hydrothermal vents at spreading centers introduce various amounts of iron, sulfur, manganese, silicon and other elements into the ocean, some of which are recycled into the ocean crust. Helium-3, an isotope that accompanies volcanism from the mantle, is emitted by hydrothermal vents and can be detected in plumes within the ocean.[7]
Spreading rates on mid-ocean ridges vary between 10 and 200 mm/yr. Rapid spreading rates cause increased basalt reactions with seawater. The magnesium/calcium ratio will be lower because more magnesium ions are being removed from seawater and consumed by the rock, and more calcium ions are being removed from the rock and released to seawater. Hydrothermal activity at ridge crest is efficient in removing magnesium.[8] A lower Mg/Ca ratio favors the precipitation of low-Mg calcite polymorphs of calcium carbonate (calcite seas).[6]
Slow spreading at mid-ocean ridges has the opposite effect and will result in a higher Mg/Ca ratio favoring the precipitation of aragonite and high-Mg calcite polymorphs of calcium carbonate (aragonite seas).[6]
Experiments show that most modern high-Mg calcite organisms would have been low-Mg calcite in past calcite seas,[9] meaning that the Mg/Ca ratio in an organism's skeleton varies with the Mg/Ca ratio of the seawater in which it was grown.
The mineralogy of reef-building and sediment-producing organisms is thus regulated by chemical reactions occurring along the mid-ocean ridge, the rate of which is controlled by the rate of sea-floor spreading.[8][9]
Human impacts
Marine pollution
Marine pollution occurs when substances used or spread by humans, such as industrial, agricultural and residential waste, particles, noise, excess carbon dioxide or invasive organisms enter the ocean and cause harmful effects there. The majority of this waste (80%) comes from land-based activity, although marine transportation significantly contributes as well.[10] Since most inputs come from land, either via the rivers, sewage or the atmosphere, it means that continental shelves are more vulnerable to pollution. Air pollution is also a contributing factor by carrying off iron, carbonic acid, nitrogen, silicon, sulfur, pesticides or dust particles into the ocean.[11] The pollution often comes from nonpoint sources such as agricultural runoff, wind-blown debris, and dust. These nonpoint sources are largely due to runoff that enters the ocean through rivers, but wind-blown debris and dust can also play a role, as these pollutants can settle into waterways and oceans.[12] Pathways of pollution include direct discharge, land runoff, ship pollution, atmospheric pollution and, potentially, deep sea mining.
The types of marine pollution can be grouped as pollution from marine debris, plastic pollution, including microplastics, ocean acidification, nutrient pollution, toxins and underwater noise. Plastic pollution in the ocean is a type of marine pollution by plastics, ranging in size from large original material such as bottles and bags, down to microplastics formed from the fragmentation of plastic material. Marine debris is mainly discarded human rubbish which floats on, or is suspended in the ocean. Plastic pollution is harmful to marine life.Climate change
Increased carbon dioxide levels, mostly from burning fossil fuels, are changing ocean chemistry. Global warming and changes in salinity[13] have significant implications for the ecology of marine environments.[14]
Acidification
Ocean acidification is the ongoing decrease in the pH value of the Earth's oceans, caused by the uptake of carbon dioxide (CO2) from the atmosphere.[15][16] The main cause of ocean acidification is human burning of fossil fuels. As the amount of carbon dioxide in the atmosphere increases, the amount of carbon dioxide absorbed by the ocean also increases. This leads to a series of chemical reactions in the seawater which has a negative spillover on the ocean and species living below water.[17] When carbon dioxide dissolves into seawater, it forms carbonic acid (H2CO3). Some of the carbonic acid molecules dissociate into a bicarbonate ion and a hydrogen ion, thus increasing ocean acidity (H+ ion concentration). Between 1751 and 1996, the pH value of the ocean surface is estimated to have decreased from approximately 8.25 to 8.14,[18] representing an increase of almost 30% in H+ ion concentration in the world's oceans (the pH scale is logarithmic, so a change of one in pH unit is equivalent to a tenfold change in H+ ion concentration).[19][20]
The ocean's pH value as of 2020 was 8.1, meaning it is currently lightly basic (the pH being higher than 7).[17] Ocean acidification will result in a shift towards a lower pH value, meaning the water will become less basic and therefore more acidic.[16] Ocean acidification can lead to decreased production of the shells of shellfish and other aquatic life with calcium carbonate shells, as well as some other physiological challenges for marine organisms. The calcium carbonate- shelled organisms can not reproduce under high saturated acidotic waters.
Ocean acidification impacts many species, especially organisms like oysters and corals. It is one of several effects of climate change on oceans.Deoxygenation

Ocean deoxygenation is the reduction of the oxygen content of the oceans due to human activities[22] as a consequence of anthropogenic emissions of carbon dioxide[23][24] and eutrophication-driven excess production.[22] It is manifest in the increasing number of coastal and estuarine hypoxic areas, or dead zones, and the expansion of oxygen minimum zones (OMZs) in the world's oceans. The decrease in oxygen content of the oceans has been fairly rapid and poses a threat to all aerobic marine life, as well as to people who depend on marine life for nutrition or livelihood.[25][26][27][28]
Oceanographers and others have discussed what phrase best describes the phenomenon to non-specialists. Among the options considered have been ocean suffocation (which was used in a news report from May 2008[29]), "ocean oxygen deprivation",[30] "decline in ocean oxygen", "marine deoxygenation", "ocean oxygen depletion" and "ocean hypoxia". The term “Ocean Deoxygenation” has been used increasingly by international scientific bodies because it captures the decreasing trend of the world ocean’s oxygen inventory.[22]History

Early inquiries into marine chemistry usually concerned the origin of salinity in the ocean, including work by Robert Boyle. Modern chemical oceanography began as a field with the 1872–1876 Challenger expedition, which made the first systematic measurements of ocean chemistry.
Tools
Chemical oceanographers collect and measure chemicals in seawater, using the standard toolset of analytical chemistry as well as instruments like pH meters, electrical conductivity meters, fluorometers, and dissolved CO₂ meters. Most data are collected through shipboard measurements and from autonomous floats or buoys, but remote sensing is used as well. On an oceanographic research vessel, a CTD is used to measure electrical conductivity, temperature, and pressure, and is often mounted on a rosette of Nansen bottles to collect seawater for analysis. Sediments are commonly studied with a box corer or a sediment trap, and older sediments may be recovered by scientific drilling.
Marine chemistry on other planets and their moons
The chemistry of the subsurface ocean of Europa may be Earthlike.[31] The subsurface ocean of Enceladus vents hydrogen and carbon dioxide to space.[32]